
Просмотров: 7142
Репрезентативные публикации за июнь 2018 г.
I.O. Koshevoy, V. Sivchik, Rajib Sarker, Zong-Ying Liu, Kun-You Chung, E.V. Grachova, A.J. Karttunen, and Pi-Tai Chou, Improvement of the photophysical performance of platinum cyclometalated complexes in halogen bonded adducts, Chem. Eur. J., 2018, 24, 11475–11484, doi: 10.1002/chem.201802182
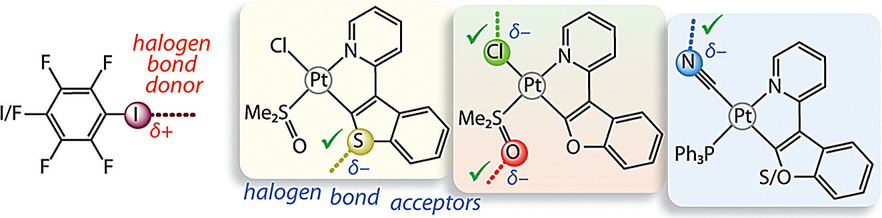
Три группы люминесцирующих платиновых комплексов [Pt(C^N)(L)(Y)] были исследованы как акцепторы галогенной связи в отношении иодфторбензолов (IC6F5 и I2C6F4). Производные фосфиноцианида (L/Y = PPh3/CN) кристаллизуются совместно как с IC6F5, так и с I2C6F4. В то время как нативные комплексы являются довольно плохими люминофорами в твердом состоянии (Φ = 0,02-0,09), аддукты проявляют до 10-кратного увеличения интенсивности свечения с незначительным изменением энергии эмиссии. Наблюдаемое усиление квантовой эффективности объясняется главным образом совместным влиянием нековалентных взаимодействий, которые регулируют режим кристаллической упаковки.
Abstract
Three groups of luminescent platinum complexes [Pt(C^N)(L)(Y)] [C^N=benzothienyl‐pyridine (1), bezofuryl‐pyridine (2), phenyl‐pyridine (3); L/Y=DMSO/Cl (a), PPh3/Cl (b), PPh3/CN (c)] have been probed as halogen‐bond (XB) acceptors towards iodofluorobenzenes (IC6F5 and I2C6F4). Compounds 1 a and 2 a (L/Y=DMSO/Cl) afford the adducts 1 a⋅⋅⋅I2C6F4 and 2 a⋅⋅⋅I2C6F4, which feature I⋅⋅⋅Sbtpy/I⋅⋅⋅πbtpy and I⋅⋅⋅ODMSO/I⋅⋅⋅Cl short contacts, respectively. The phosphane‐cyanide derivatives 1 c and 2 c (L/Y=PPh3/CN) co‐crystallise with both IC6F5 and I2C6F4. None of the phpy‐based species 3 a–3 c participated in XB interactions. Although the native complexes are rather poor luminophores in the solid state (Φem=0.023–0.089), the adducts exhibit an up to 10‐fold increase of the intensity with a minor alteration of the emission energy. The observed gain in the quantum efficiency is mainly attributed to the joint influence of non‐covalent interactions (halogen/hydrogen bonding, π–π stacking), which govern the crystal‐packing mode and diminish the radiationless pathways for the T1→S0 transition by providing a rigid environment around the chromophore.
Koronatov A.N., Rostovskii N.V., Khlebnikov A.F., Novikov M.S. Rh(II)-Catalyzed Ring Expansion of Pyrazoles with Diazocarbonyl Compounds as a Method for the Preparation of 1,2-Dihydropyrimidines, J. Org. Chem., 2018, doi: 10.1021/acs.joc.8b01228
В работе описана новая Rh(II)-катализируемая реакция диазокарбонильных соединений с 1,4-ди- и 1,4,5-тризамещенным пиразолами как первый пример внедрения карбеноида в простую связь N-N. Эта новая реакция представляет собой новый подход к 4-незамещенным 1,2-дигидропиримидинам, позволяющий осуществлять широкое варьирование функциональных групп в целевом гетероцикле. Согласно DFT расчетам, расширение пиразольного цикла протекает через последовательное образование связанного с металлом пиразолиевого илидая, свободного пиразолиевого илида и 1,5-диазагексатриена,претерпевающего 1,6-циклизацию на последней стадии домино процесса.
Abstract
A high yield synthesis of 1,2-dihydropyrimidines by the Rh(II)-catalyzed reaction of diazocarbonyl compounds with 1,4-di- and 1,4,5-trisubstituted pyrazoles is reported. This reaction represents the first example of a carbenoid insertion into a N–N bond and provides a novel approach to 4-unsubstituted 1,2-dihydropyrimidines with a broad range of functional group tolerance. According to DFT calculations, the pyrazole ring expansion proceeds via the sequential formation of the metal-bound pyrazolium ylide, metal-free pyrazolium ylide, and 1,5-diazahexatriene followed by 1,6-cyclization.).

Maria P. Sokolova, Michael A. Smirnov, Artemiy A. Samarov, Natalya V. Bobrova, Vitaly K. Vorobiov, Elena N. Popova, Elizaveta Filippova, Pavel Geydt, Erkki Lahderanta, Alexander M. Toikka Plasticizing of chitosan films with deep eutectic mixture of malonic acid and choline chloride, Carbohydrate Polymers, V. 197, 548–557, 2018, doi: 10.1016/j.carbpol.2018.06.037
Получены и исследованы пленки хитозана, содержащие глубокий эвтектический растворитель (DES) на основе малоновой кислоты и хлориде холина. Содержание DES варьировалось от 0 до 82 вес%. Анализ изотерм сорбции воды, данные атомно-силовой микроскопии и результаты Фурье ИК-спектроскопии подтвердили сильное взаимодействие хитозана с компонентами ДЭС. Пластифицирующий эффект DES отражают проведенные статические объемные механические измерения, термический анализ и количественное наномеханическое картирование (QNM). Введение DES в пленки хитозана приводит к температуре стеклования в области +2 - 2.3 °C. Результаты QNM подтверждают однородность пленок, содержащих до 75 мас.% DES.
Abstract
Chitosan (CS) films containing deep eutectic solvent (DES) based on malonic acid (MA) and choline chloride (ChCl) were successfully prepared by solution casting method by using DES content ranging from 0 to 82 wt%. A strong interaction of CS with the components of DES was demonstrated by analyses of water sorption isotherms, atomic force microscopy and FTIR results. The plasticizing effect of the MA and ChCl mixture on the CS matrix was shown by static bulk mechanical measurements, thermal analysis and quantitative nanomechanical mapping (QNM). Elongation at break increased from 3 to 62% at increase of DES content from 0 to 67 wt%, while further increase of DES content led to the decreasing of maximal elongation. Introduction of DES into CS films led to the appearance of glass transition temperature in the region +2 - -2.3 °C. QNM results indicated homogeneity of the films containing up to 75 wt% of DES.



